Mordants are indispensable in histological microtechnique. The most common staining methods would not be possible without them, the H&E is a good example. Despite their importance there is sometimes some confusion as to what mordants are, or are not. The term is often applied to agents that are, in fact, not true mordants.
What is a Mordant?
A mordant is a metal with a valency of at least two. The two commonest metals used in histotechnology are aluminum and ferric iron, both with valences of three. The attachment of mordants to dyes is by means of a covalent and a coordinate bond. This is otherwise known as chelation, and is a relatively common phenomenon. Stopping blood clotting by removing calcium ions with EDTA is an example. The word chelation is apparently derived from the name of the large claw, or chela, of a lobster. Gripping a metal atom by two different bonds has a fanciful similarity to gripping prey with two parts of this claw.
A mordant can be defined as a polyvalent metal ion which forms coordination complexes with certain dyes.
Mordant Criteria
Mordants are metals. However, not all metals are mordants. The most common mordant metals in histotechnology are aluminum and ferric iron, both with valences of three. Less commonly, other metals may be used as mordants, such as beryllium, calcium, chromium, lead, tungsten, molybdenum etc. By definition, however, a mordant must be a metal. If a chemical is not metallic, it is not a mordant.
Note that iodine is not a metal. Therefore it is not a mordant. It’s function in Gram’s stain is as a trapping agent. Similarly, the use of Bouin’s fluid before Masson’s trichrome is an example of secondary fixation of sections. Picric acid is not a metal and consequently is not a mordant. Bouin’s fluid contains no metal ions, and therefore cannot mordant tissues. It can, and does, refix them to make them more avid for acid dyes. This is a well known characteristic of picric acid fixation, used to good effect here.
What is a Lake?
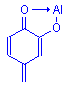
The chelate formed from a mordant dye and a metal is called a lake. This term is derived from lac, a mordant dye obtained from an insect in India, and from which shellac is obtained. Over time the term lac has changed to lake and has come to be the generic term for all dye-mordant complexes.
A lake can be defined as a coordination complex formed between a polyvalent metal ion and certain dyes
How Lakes Form
Two types of bonds are involved in the fundamental reaction between a mordant dye and a mordant. One is a covalent bond with a hydroxyl oxygen. The other is a coordinate bond with another oxygen (the electron donor).
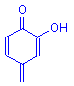
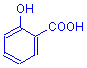
In the table below are the four most common configurations found in mordant dyes. There are others, particularly in the textile industry, but nearly all mordant dyes used in biological staining have one or more of these configurations. Note that in each case a double bonded oxygen and a hydroxyl group (or a carboxyl group) are involved.
The covalent bond forms between the hydroxyl oxygen and the metal.
The coordinate bond forms between the double bonded oxygen and the metal.
Structure

Lake

Structure
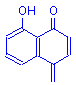
Lake

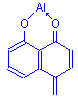
Since aluminum and ferric iron both have valencies of three, it is possible that three molecules of dye could attach to each atom of the mordant metal. In practice, it is unlikely that this happens, since attachment to the tissue is also by means of the mordant metal. Varying the amount of mordant present with the dye is a way to exert some control over the staining characteristics of some lakes. This is quite effective with alum hematoxylin solutions. Regressive formulations usually have larger amounts of dye present in comparison to progressive formulations. In other words, the amount of mordant available to each dye molecule is lower.
It is possible, however, that the formation of dye-mordant complexes involving several molecules of dye can form. This could occur if a dye had more than one chelating point, such as carmine, and was able to form multi molecule – multi chelate complexes. Such large complexes may explain some staining characteristics of particular lakes, e.g. the selectivity of mucicarmine for mucins.
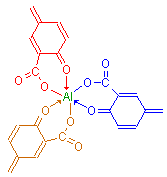
Triple chelate
It is often remarked that the addition of a mordant to an appropriate dye solution results in a very sudden, dramatic change in color. This is due to the incorporation of the metal atom into the delocalised electron system of the dye. Metals have relatively low energy levels, so their incorporation into a delocalised system results in a lowering of the overall energy. The absorbance of the lake, and thus its color, is related to this.
How Mordants Attach To Tissue
The attachment of the mordant metal to tissue is essentially by the same mechanism as that causing attachment to the dye. That is by chelation – covalent and coordinate bond formation.
Phosphate hydroxyl groups of the nucleic acids provide means for covalent bonding, and other atoms in the vicinity can donate electrons for the coordinate bond. The DNA strand has a repeating sequence of phosphate and deoxyribose with a base attached, the bases pairing up in a complementary manner of guanine with cytosine, and adenine with thymine, and forming the double helix. For histological staining the bases are not significant, but the phosphate groups are fundamentally important. Attachment of mordanted dye is due to the mordant forming a chelate with a phosphate hydroxyl and another atom in a manner very similar to that between the mordant and the dye. Although the diagram below shows the source of electrons for the co-ordinate bond to be also a phosphate oxygen, this is for convenience only.
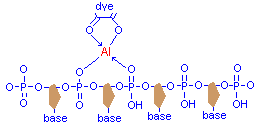
It is a similar case with proteins, as there are many hydroxyl and carboxyl groups available. It should be noted that nuclear chromatin contains both protein components and DNA, so that two seperate staining events may be happening at the same time. That is why some staining methods may still demonstrate nuclear structure even after all DNA has been removed.
Using Lakes
Mordants and dyes may be applied in three ways. The terms used are defined below. The suffix -chrome in these terms refers to chromium, which is a common mordant in textile dyeing. The terms are borrowed from that industry. In histotechnology chromium is only rarely used, but the terms are still used.
Onchrome
- The mordant is applied first, followed by the dye.
- Heidenhain’s iron hematoxylin is a classic example.
Metachrome
- Mordant and dye are mixed together then applied.
- Alum hematoxylin solutions are applied like this. It is probably the commonest way to use mordant dyes in histotechnology.
Afterchrome
- The dye is applied first, followed by the mordant.
- This is hardly ever done in histotechnology, although Lillie used the obscure dye phenocyanin TC followed by a ferrous mordant in this way.
References
- Baker, John R., (1958)
Principles of biological microtechnique
Methuen, London, UK.






