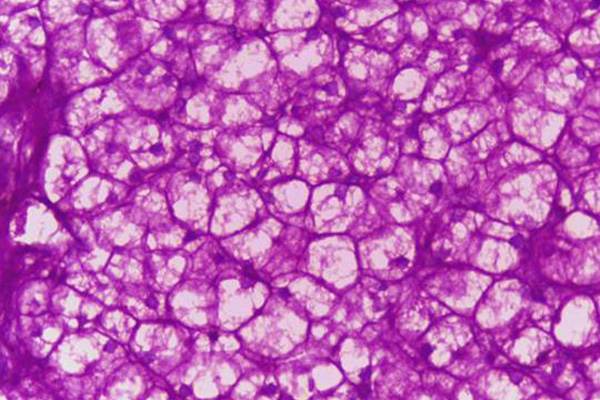
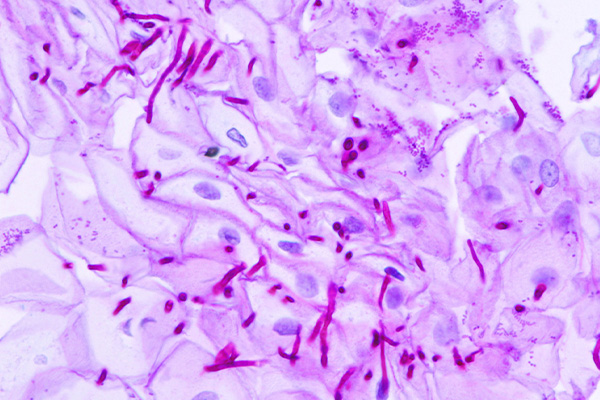
Periodic Acid Schiff Reaction
The periodic acid Schiff reaction (PAS) is used to demonstrate the presence of 1-2-glycols, and is consequently an important method in the histochemistry of carbohydrates and the histological demonstration of many structures.
Expected Results
- 1-2-glycols – red or dark purple
- Nuclei – blue
Materials
- Periodic acid – 1% aqueous
- Schiff’s reagent
- Mayer’s hemalum
Tissue Sample
5µ paraffin sections of neutral buffered formalin fixed tissue are suitable. Other fixatives are usually satisfactory, although glutaraldehyde should be avoided.
Protocol
- Bring sections to water via xylene and ethanol.
- Place into periodic acid for 10-30 minutes.
- Rinse well with tap water.
- Rinse with distilled water.
- Place in Schiff’s reagent for 10-30 minutes.
- Wash off with distilled water.
- Wash well with tap water for about 10 minutes.
- Counterstain with Mayer’s hemalum for 2 minutes.
- Wash well with tap water until hemalum is blued.
- Dehydrate with ethanol, clear with xylene and coverslip using a resinous medium.
Notes
- Glutaraldehyde fixation leaves free aldehyde groups attached to tissues, which causes an overall positive reaction. These groups may be stopped from reacting with an appropriate procedure such as the aniline-acetic aldehyde block.
- The tap water wash at step 7 is necessary to develop the red color. Within limits, the longer the wash the darker the color.
- Originally, it was recommended that the Schiff’s reagent be washed off with dilute sulfurous acid (the sulfite rinses). Since water recolors Schiff’s reagent, it was believed that a water wash could lead to false positive results. It is now known this is not the case, provided the Schiff’s reagent is removed quickly and the sections do not stay in water contaminated with it for extended periods.
Safety Note
Prior to handling any chemical, consult the Safety Data Sheet (SDS) for proper handling and safety precautions.
References
- Culling C.F.A., (1974)
Handbook of histopathological and histochemical techniques Ed. 3
Butterworth, London, UK.