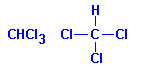
Chloroform, also known as trichloromethane and methyl trichloride, does not raise the refractive index of tissues as it replaces the dehydrant. It still gives a visual cue since tissues float with most of the tissue above the surface of the fluid when first applied. The tissues then sink down as the dehydrant is replaced and, when it is complete, the majority of the tissue will be below the surface of the chloroform. Texts often say the tissue sinks, but this is not in the sense that it falls to the bottom of the container, but in the sense that the upper surface is at the fluid-air interface instead of the lower surface at the interface, as it is initially.
It has a distinctive odour which not everyone finds unpleasant. It is a quite gentle clearing agent and causes very little damage. It shrinks tissue hardly at all and does not cause appreciable hardening. Unfortunately, it has some drawbacks. It is, of course, an anesthetic and fumes in a confined space may potentially be hazardous. It does cause liver damage if inhaled repeatedly, and that is unavoidable during processing. It can, under the right circumstances, produce phosgene, a poisonous gas used during WWI. These drawbacks are, however, more potential than actual when normal laboratory safety precautions are used. It is a useful clearant for special cases such as decalcified hard bone, which will be left relatively soft after infiltration.
One serious disadvantage is that chloroform attacks rubber and plastics so it cannot be used on most modern automatic tissue processors, since the seals will be destroyed. It is suitable for use on Histokine and Auto-Techncon style rotary processors.
Carbon Tetrachloride
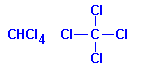
Carbon tetrachloride (tetrachloromethane) is similar to chloroform. It is not inflammable but it is toxic and causes liver damage. It also forms phosgene on heating. It is not recommended.
Carbon disulphide
Although it has an unpleasant odour, likely caused by contamination with carbonyl sulfide, carbon disulfide is an efficient clearing agent. It is very volatile, and quite inflammable, which, together with its penetrating odour make it completely unsuitable for automatic processing machines whether of the newer or older styles. For manual processing of a rapid specimen it can be useful. It removes ethanol rapidly and is itself rapidly removed by the hot wax. In fact, when placed into the wax, bubbles of vaporizing carbon disulfide can be seen rising from the specimen. Care has to be exercised with its use, but it is otherwise reasonably useful on occasion. The top formulae are carbon disulfide, the bottom formulae are of the contaminant, carbonyl sulphide, the source of the rotten cabbage smell.






