One of the most fundamental principles in the preparation of high quality slides for microscopic examination is that fixation is the foundation on which all subsequent procedures build. If this step is not carried out effectively, then it is not possible to rectify it at a later date and what may be done with the fixed tissue will always be limited by what was done initially.
Introduction to Fixation
The purpose of fixation is to preserve the tissue in such a way that it appears the same in a microscopic preparation as it appeared while living and functioning. Of course, we must acknowledge that this is an impossible aim, as the very act of fixation stops all functions and distorts in some fashion the appearance. Nevertheless, the aim should be to approach this ideal as closely as possible and many combinations of chemicals have been recommended for it with varying degrees of success.
Fixatives not only preserve tissues, they chemically alter them in some fashion, usually due to denaturing their proteins. This can be by precipitation, as with ethanol, or by forming compounds with them, as formaldehyde. In both cases the proteins are preserved in situ with little distortion, thus keeping the tissue structure intact. There may be effects on carbohydrates and lipids as well, but it is the proteins which are the main target.
Actually fixing carbohydrates and lipids, as in chemically altering and rendering them inert, is usually considered to be a specialist procedure requiring separate treatment. An exception is when carbohydrates and lipids are in the form of complexes with proteins, glycoproteins and lipoproteins for instance, when fixation can occur via the protein component. In other cases carbohydrates are simply trapped within a meshwork of fixed protein. Lipids may be unaltered and simply dissolve out during processing.
When properly and thoroughly fixed, blocks of tissue should be able to resist the effects of other reagents used during the processing procedure. In particular, resistance to the effects of ethanol is important as it is capable of fixing proteins in its own right. If the tissue is not properly fixed before the ethanol for dehydration is applied, there is a distinct likelihood that it will complete the fixation while dehydrating. Unfortunately, when used alone as a fixative, ethanol is quite poor, causing shrinkage and making tissues hard and brittle. One side effect of the modern minimalist approach to fixation and processing, in which formalin is applied barely long enough to stabilize the proteins but certainly not long enough to cross link them, is that ethanol fixation during processing has become fairly commonplace. In fact, this is so common that it has been referred to as “parched earth” artifact, or “overfixation”, a rather ironic term considering that it is caused by not fixing the tissue long enough in formalin. The resolution to this problem is to fix tissues thoroughly before processing. If there is insufficient time for this to be done with formalin, then another fixative that can do it should be used.
Any microorganisms present in the tissue are likely to be killed and preserved along with the tissue, although there may be a few isolated types of bacteria which can resist this treatment. Similarly, the enzymes that cause autolysis are usually rendered inactive, either through denaturation because they are themselves proteins or because their substrate, cellular components and contents, have been denatured. In either case, putrefaction and autolysis come to an end. Once the tissues have been removed from the fixative, however, microorganisms in the environment may colonize the tissue and putrefaction may begin again unless precautions are taken against it. These precautions include the completion of processing. It is generally accepted that no bacteria can resist the complete process from fixative to paraffin infiltration, and paraffin processed tissues are considered to be safe to handle and stable indefinitely. It is known that prions can resist this complete process, so precautions must be taken to ensure personal safety if the tissue is from this source.
One of the observed effects of fixation is that the tissue tends to harden somewhat, the degree being dependent on which specific chemicals are used to fix. In moderation this is of benefit as it tends to even out the differences in hardness of the individual components and layers, and this helps during sectioning. It can be overdone, of course, and excessive hardening is a detriment. Often, excessive hardening is due to the tissue being left too long in the fixative, particularly if harsh precipitant chemicals are used. Fortunately, the hardening from formalin is relatively mild.
Tissues may also become brittle, which can make sectioning very difficult as the tissues may shred and shatter as the sections are cut. Sectioning much thinner, cooling the block and soaking the tissue in ice cold water may all help when this is encountered. It is more prevalent with areas of proteinaceous exudates, colloids and similar such materials, but it can occur in many tissues with harsh fixatives.
Most fixatives cause a degree of shrinkage, or give inadequate protection to the tissue so that shrinkage occurs during dehydration and clearing. It can be easily seen that most shrinkage occurs during the infiltration step in hot wax. It can be quite striking to observe the first wax bath during the first 30 minutes of application as the tissue diminishes in size quite rapidly. Shrinkage not only occurs overall with the piece of tissue becoming smaller in dimensions. It also occurs at a cellular level, and cells and fibers may separate from each other introducing spaces where non existed during life. Simple formalin solutions permit significant shrinkage even when the tissue is thoroughly fixed. Harsher, protein precipitant solutions tend to resist it more.
Practicalities
It cannot be stressed enough that tissues should be thoroughly fixed before proceeding to the dehydration, clearing and infiltration steps. Unfortunately, in the quite understandable rush for quick diagnoses this basic principle has often been forgotten, with a consequent reduction in quality. Most often this shows with the use of simple formalin mixtures, probably the most commonly used fixative, which is often carried out for an inadequate length of time, allowing subsequent treatments with harsh reagents to modify the microscopic appearance. Formalin on its own requires at least overnight at room temperature, and preferably several days, but it is often applied for just a few hours or even less. Frequently, this is accompanied by subsequent complaints of poor quality sectioning, staining and appearance.
Under these circumstances it would make more sense to use a faster fixative so that fixation is complete within the available time. This may not be possible in practice as many of the fixatives which would be useful for this contain mercuric chloride, a chemical which may no longer be used in some jurisdictions or whose use is deprecated even if not under outright ban. As well, the modern use of immunological methods requires a fixative that permits various antigens to be identified in fixed tissue, and the characteristic of formalin that allows cross linking of proteins to be reversed by moist heat (HIER) is useful. When sufficient tissue is available one sample may be processed as quickly as possible while a second is fixed more thoroughly and then processed, but small biopsies often do not permit this. On occasion, increasing the temperature of the formalin to about 50°C or 60°C will usually speed up fixation significantly, but this is not a panacea as there is a reduction in the quality of preservation and there may be consequences from the heat itself, which can denature proteins by cooking.
The best resolution, from a technical perspective, is patience. Chemical reactions take some time to complete, and fixation is no different than any other chemical reaction. It is strongly recommend that sufficient time be allowed for the fixative to fix, and that processing be delayed until that is so. It causes only a short delay in diagnosis, and for those specimens where a very rapid diagnosis is required frozen section diagnoses may be used.
A sometimes overlooked factor in fixation is the amount of fixative used. There must be enough to permit free access to all parts of the tissue. Cramming a large piece of tissue into a small container, then pouring a little fixative over it is just not good enough, but it is frequently encountered. The tissue must be able to float freely in the fixative and there should be not less than 10 times the volume of tissue, and preferably about 20 times the volume.
If the piece of tissue is large enough to require a long time for the fixative to penetrate to the center, then it should be described and “breadloafed”, or cut into slices no thicker than 1 cm. This will allow the fixative to penetrate more effectively. Intestines should be opened and any contents removed. It may then be replaced into a large volume of fixative, or if small enough, pinned out with plastic pins (to avoid corrosion) and floated in the fixative.
Chemical Fixing Agents
The number of chemicals used for histological fixation is fairly limited, with some being more popular than others, and some hardly used at all any more for various reasons. Any chemical which can preserve the structure or chemical composition of animal or plant tissues is considered to be a fixing agent.
In order to accomplish this preservation, fixing agents are primarily effective against the proteins in the tissue. In some way each agent must denature, or chemically alter, these proteins. In the process the individual agents may, or may not, combine with the proteins (or other tissue components). Likewise they may, or may not, precipitate (or coagulate) the proteins as a consequence of their fixing action.
Agents which combine with proteins are called additive, and those which do not are called non-additive. Agents which precipitate proteins are called coagulant, and those which do not are called non-coagulant.
By combining these characteristics it is possible to classify fixing agents into four groups:
- Additive coagulant
- Additive non-coagulant
- Non-additive coagulant
- Non-additive non-coagulant
The terms coagulant and precipitant are synonymous in this context as they both refer to precipitation of proteins and coagulation of protein gels.
This classification is of limited usefulness as there is little to no effect on the handling of tissues, nor of the results obtained, whether a fixing agent is attached to the tissue or not or whether proteins are precipitated or not. The exception is chromium as it can be used as a mordant, so fixation with chromic acid or potassium dichromate may be useful for demonstrating some tissue components with a mordant dye.
| Fixing agent | Additive | Coagulant |
|---|---|---|
| Acetic acid | No | No |
| Acetone | No | Yes |
| Chromium Acid | Yes | Yes |
| Ethanol | No | Yes |
| Formaldehyde | Yes | No |
| Glutaraldehyde | Yes | No |
| Mercuric chloride | Yes | Yes |
| Methanol | No | Yes |
| Osmium tetroxide | Yes | No |
| Picric acid | Yes | Yes |
| Potassium dichromate | Yes | No |
| Trichloracetic acid | Yes | Yes |
| Zinc Chloride | Yes | Yes |
Other Fixing Agents
Other than the standard fixing agents discussed in other pages, there have been some used in the past which are now obsolete, or nearly so. There is no consistent reason for that, perhaps it was the cost, the corrosive nature of the chemical, ease of use and so on. Many of these still have their advocates, and they may very well perform admirably.
Copper salts are sometimes still spoken of as being very effective for nuclear preservation. The copper in fixatives which contain it may act as a mordant, in the true sense, for some dyes including hematein. This, of course, infers that copper is an additive fixing agent.
Platinum chloride was in use at the beginning of the 20th century, but its use never caught on since it is very expensive. The chemical actually used was chloroplatinic acid, which bears much the same relationship to platinum chloride as chloroauric acid does to gold chloride, that is, a convenient but inaccurate name.
In addition, some other heavy metals have been used on occasion, as have some aldehydes and alcohols. In many cases these chemicals have no particular advantage over those we usually employ, and may be more difficult to work with as being more expensive, toxic, inconvenient etc. Nevertheless, it can be interesting to investigate them for self-education. Should you do so, please use them and dispose of them safely.
How do I Choose a Fixative?
Fixative Properties
A particular fixing agent may be better at preserving some tissue constituents than it is at preserving others. Fixative mixtures that contain this agent may be a better choice for fixation when this constituent is the target for examination.
An example would be acetic acid, which is excellent for nucleoprotein. Fixatives that contain acetic acid often give sharp and clear preservation of nuclear structure as a consequence. For that reason an acetic acid fixative would be a good choice when nuclear structure is important.
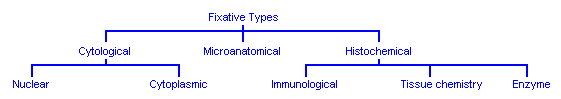
Fixatives are often classified according to whether they preserve structures in relation to each other, or whether they preserve individual cellular constituents.
Those fixatives that target overall tissue structure are called microanatomical fixatives. Those that target cellular components are called cytological fixatives. Of this group, some are nuclear preservatives in contrast to others which are far better at cytoplasmic preservation. Some other fixatives target particular chemicals in the tissue, including enzymes and antigens. These are the histochemical fixatives.
No classification system for fixation effects can be considered to be absolute as fixatives may be satisfactory for more than one purpose. As an example, formal sublimate is considered to be primarily a microanatomical fixative but it also preserves nuclear structure well, and formalin fixed tissues are often used for immunological staining.
Index of Fixatives
Find details on the chemical properties, preparation, treatment, and more of over 40 different fixatives.
Fixation Artefacts
Artefacts (or artifacts) are human made items or the results of human activity. Fixation artefacts are consequently changes brought about in the tissue as a result of human activity, i.e. putting some tissue in a fixative.
From that definition, fixation itself could be considered to be an artefact, as it is a change brought about by human activity, and sometimes that comment is made. It is certainly true that fixation alters the tissue state: it ceases to be a dynamic system and becomes static, with most chemical processes and physical structures suspended in the state they were when the fixative’s chemicals encountered them. Despite that, a lot can be determined by examining that snapshot in time which is represented by a microscope section.
More particularly however, artefacts are usually defined as the unwanted effects of a process, including fixation. In practice these unwanted effects are largely restricted to a few items such as deposits from the fixative or its reactions with tissue components, and the physical effects of fixation we would prefer to do without, such as detectable shrinkage, both at the gross and microscopic levels; the hardening of some tissues which causes chatters parallel to the knife edge; and the brittleness from some fixatives which results in shattering and cracking during sectioning.
There are two deposits in particular that we may encounter fairly often. Formalin pigment, or acid formaldehyde hematein, is frequently seen as fine granules, particularly in bloody tissues, and mercury pigment is invariably encountered in tissues fixed with mercuric chloride. Both are easily removed.
Although, strictly speaking, it is not a fixation artefact, the removal of triglycerides – fats – is a consequence of the failure of the fixative to render them unaffected by subsequent reagents. Most fixatives are deficient in this area, and blank holes are often seen where there was, in life, fat globules.
Not too many years ago a new artefact began to be mentioned, and was named the parched earth effect. It quickly became apparent that this was not a previously undetected artefact, but was, in fact, the undesirable effects of warm ethanol fixation. It began to be seen when more laboratories switched to processing times of less than 24 hours from biopsy to diagnostic report. This can only be done with paraffin sections if each step is cut to the absolute minimum. Unfortunately, many laboratories did not give adequate time for the most fundamental step, fixation, to be adequately effective and protect the tissue. The result was that fixation was completed in subsequent steps with (warmed) ethanol. Parched earth artefact, then, is just the poor quality fixation obtained with ethanol, exaggerated by applying it warm to speed up dehydration.
Learn more about these 3 different fixation artefacts below.
Secondary Fixation
Secondary fixation is the term used for the practice of initially fixing with 10% formalin, then refixing with another fixative. The second fixative refixes the tissue so that some of its characteristics can be obtained. In this way it is possible to obtain fixation with different characteristics on different blocks from the same bulk tissue. One advantage of this procedure is that it can be applied to tissues that have been fixed with a formalin variant and stored in the fixative for some time.
The secondary fixative chosen is usually a strong precipitant type which partially overcomes the protective effect of formalin. It must be emphasized, however, that secondary fixation does not give the same results as would have been obtained if the secondary fixative had been applied to fresh, unfixed tissue. It must surely be self evident that only those structures and organelles preserved by the primary fixative can be affected by the secondary fixative. If the structure or organelle is no longer there, no procedure can restore it. Secondary fixation is therefore not a panacea for improper fixative selection initially.
The most popular secondary fixatives are often those that contain mercuric chloride, such as formal sublimate or Helly’s solution, although the procedure is not confined to those. This makes the practice more difficult, since all materials must be collected and disposed of properly. Fixatives which do not contain mercury can still be used with reasonable effect.
Post Chromation
Post chromation may also be referred to as post chroming or post chromatization. It is the practice of treating tissues fixed in a primary fixative, usually one containing formalin or potassium dichromate, with a simple solution of potassium dichromate for an extended period, usually days and sometimes weeks. The purpose is to improve the preservation of lipid and lipoprotein materials that would otherwise be removed by processing reagents. It is not absolute in its effect, but does improve the preservation of material such as mitochondria and myelin so they can be demonstrated in paraffin sections.
The process does not well preserve triglycerides and, for them, frozen sections or post osmication should be used.
Fixed tissues are placed into a 2.5% to 3% solution of potassium dichromate in distilled water and put into the dark. Potassium dichromate may be photosensitive and the absence of light is desirable. It is left for the required time, often a few days to a week, then washed with running tap water overnight to remove all remaining dichromate. The tissue may then be processed.
It will undoubtedly be found that the tissue is brittle. This is inherent in the process and the degree tends to increase as the duration of post chroming lengthens.
This process is not just a fixation step, but it may also mordant the tissues in the true sense of the term. Dichromate contains chromium which is an effective mordant for some dyes. It may be retained in conjunction with some lipid material and be used to demonstrate them. This is particularly so with myelin.
Culling points out that sections may also be post chromed by placing dewaxed sections into a dichromate solution for 12 to 24 hours, followed by washing and staining. Of course, material that is removed during processing will no longer be susceptible to post chroming, although surprising amounts of protein bound lipids do resist extraction and are still present.
Post Osmication
The process of post osmication is to preserve lipids so that they may be seen in paraffin sections. Unlike post chromation, post osmication allows the demonstration of all lipids, including triglycerides. Staining is usually unnecessary as the post osmication procedure itself blackens lipids, and they will appear darkly coloured in unstained paraffin sections. Many dyes stain poorly following osmium treatment.
- Following fixation in simple formalin for at least 24 hours, wash thin slices of tissue overnight in running tap water to remove all traces of formalin.
- Soak the washed tissue in a few changes of distilled water for one hour or longer.
- Place the tissue into 0.5% osmium tetroxide in distilled water for 24 hours.
- Soak the washed tissue in a few changes of distilled water for one hour or longer to remove excess osmium tetroxide.
- Complete the removal of osmium tetroxide by washing in running tap water overnight.
- Either process immediately via dehydration, clearing and infiltration, or place into a processing cassette, and store in a simple formalin variant until routine paraffin processing is done. Note that formalin will reduce any osmium tetroxide in the tissue and make it appear black.
- Section the tissue block, and either remove paraffin wax with xylene and coverslip unstained, or stain by an appropriate procedure.
- Sections coverslipped without staining will show all lipids as black or grey, including triglycerides and lipoproteins.
Notes
Osmium tetroxide is very expensive, so it is suggested that 0.1 gram sealed vials be purchased for single occasion use and the contents of one vial dissolved in 20 mL distilled water. The chemical must be dissolved in distilled water as it is very susceptible to reduction. It is safest if triple distilled water is used but that is not absolutely necessary. Unused solution may be kept for a short while if light is excluded, but any form of contamination will cause reduction and render it useless.
Osmium tetroxide gives off fumes which cause a piercing headache. Avoid inhaling these fumes. Make the solution in a fume chamber which is turned on and extracting properly. Carry out the procedure under the fume chamber until the final wash. Dispose of the used osmium tetroxide solution and washings according to local regulations.
Fixation and Staining
Fixation is not usually considered to be involved in explanations as to how dyes stain tissues. In reality, of course, fixation is intimately involved in staining. The reactions that occur during any staining process must involve tissue constituents in some fashion. So their preservation and treatment prior to staining must be factors to consider. Fixation, dehydration, clearing, and to a lesser degree, infiltration, may have an impact on the final product. However, the greatest impact from preparatory treatments undoubtedly comes from the chemicals used to denature the proteins during fixation.
It is as well to stress that the fundamental means of tissue preservation is to alter protein in some fashion. Unfixed tissues are quite avid for dyestuffs, as shown by the reduced staining times required for frozen sections with H&E. The overall effect of most fixative chemicals is to reduce this avidity to a degree. Some reduce it very little, some a lot.
We usually do not view it from this perspective. Although we know that formalin fixation reduces dye uptake, we think of fixation in neutral buffered formalin as the standard. To a large degree, in common practice, it has become the base line for reference. For that reason, we often rate other fixative chemicals in relation to the staining and preservation characteristics obtained with formalin. There is nothing wrong, or undesirable in this particularly. One base line for reference is just about as good as any other. We should keep in mind, however, that fixation does not usually increase dye uptake, rather it tends to diminish it.
Staining is so intimately associated with the preparatory techniques used that it would be foolish to discuss one without commenting on the other. While understanding the underlying principles involved in staining can be used to manipulate the process effectively, part of what must be understood is the effects of fixatives which may be used. These can have a very distinct impact on how a dye may stain a particular tissue.
There are numerous examples that can be given. No one who wanted to have good H & E staining of routine sections would use osmium fixation. Quite apart from the cost, the eosin staining would be terrible. On the other hand, it would be most appropriate to use an acetic acid containing fixative if the primary target for demonstration was the cell nucleus. With cytoplasmic granules as the primary target, it should probably be avoided.
It is the histotechnologist’s responsibility to be aware of the most appropriate fixative to use, to know what staining techniques are most effective following fixation in particular mixtures, and to be aware of the likely results of unusual fixative and staining technique combinations.
References
- Culling C.F.A., (1974)
Handbook of histopathological and histochemical techniques, Ed. 3
Butterworth, London, UK. - Baker, John R., (1958)
Principles of biological microtechnique
Methuen, London, UK. - Gray, Peter. (1954)
The Microtomist’s Formulary and Guide.
Originally published by:– The Blakiston Co.
Republished by:– Robert E. Krieger Publishing Co. - Drury, R.A.B. and Wallington, E.A.,
Carleton’s Histological Technique, Ed. 5,
Oxford University Press, Oxford, UK.






