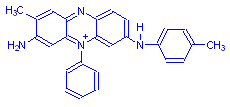
Mauveine A
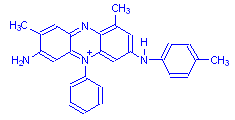
Mauveine B
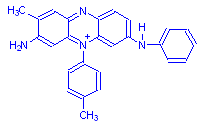
Mauveine (Gurr)
Mauve Aniline purple Perkin’s violet
Description
This dye is not used in histology but is an important compound historically, as it was the first synthetic dye to be made from aniline. Its accidental preparation by a student, named William Henry Perkin, eventually led to the modern chemical dye industry, thus indirectly to the way we practice histotechnology today.
In 1967, Edward Gurr was given a small sample of mauveine which was 111 years old at the time, i.e. produced in 1856 the same year Perkin first made it. The information in the chart above is based on that sample, and on the structural formula as given by Gurr, above. Due to the small amount of dye available, the data is inconclusive.
The structural formula was fully determined in 1994, and it was shown that mauveine is actually a mixture of two compounds, mauveine A and mauveine B above. Instructions have also been published so that the dye can be made in small quantities (Scaccia, Coughlin and Ball).
References
- Edward Gurr, 1971
Synthetic dyes in biology, medicine and chemistry
Academic Press, London, England. - Gurr, E.,
Perkin’s original alizarin and mauveine after 100 years and more.
Journal of the Society of Dyers and Colourists, v. 85, p. 473-474 - Gurr, E.,
Spectroscopy: Old stains examined.
Nature, London. v. 224: 5224, p. 1061-1062. - Meth-Cohn, O. and Smith, M.
What did W. H. Perkin actually make when he oxidized aniline to obtain mauveine?
Journal of the Chemical Society, Perkin Transactions, 1994, p. 5. - Scaccia, R. L., Coughlin, D. and Ball, D. W.,
A microscale synthesis of mauve.
Journal of Chemical Education, v. 75, p. 769.






